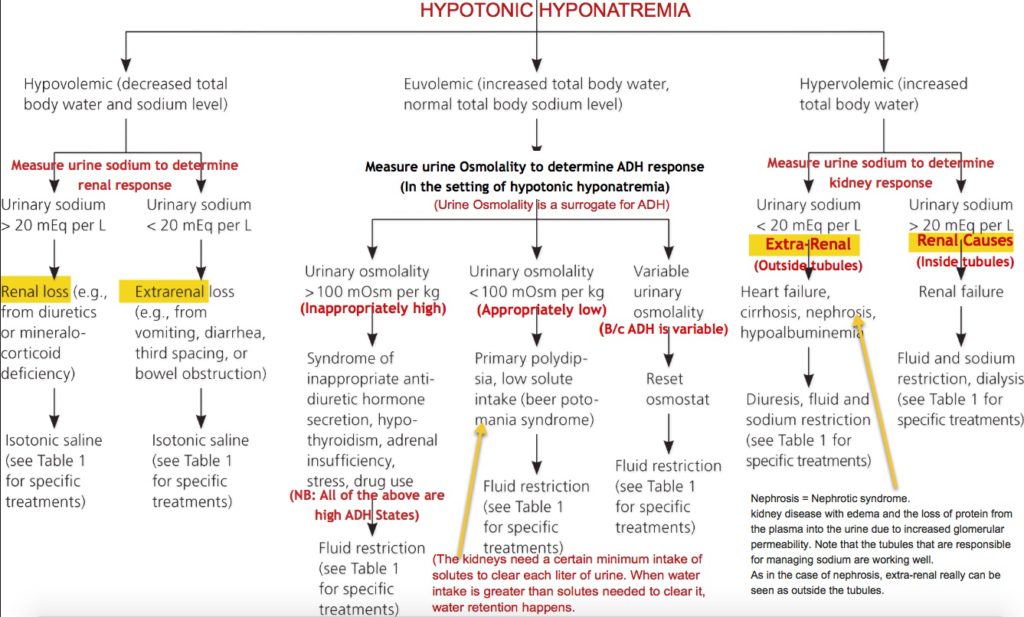
It is especially useful in helping to differentiate the cause of a metabolic acidosis, as well as following the response to therapy. The anion gap provides an estimation of the unmeasured anions in the plasma and is useful in the setting of arterial blood gas analysis. Question: Please define the Anion Gap and its utility in diagnosis and how it relates to osmolality. Abdominal pain may be a symptom of ketoacidosis or part of the inciĬommon Laboratory (lab) Values - Anion Gap Metabolic states in which acidosis is predominant include lactic acidosis and ingestion of drugs such as salicylates and methanol. Common problems that produce ketosis include alcoholism and starvation. The primary differential diagnosis for hyperglycemia is hyperosmolar hyperglycemic state (Table 23,20), which is discussed in the Stoner article21 on page 1723 of this issue. The conditions that cause these metabolic abnormalities overlap. The most common precipitating factor is infection, followed by noncompliance with insulin therapy.3 While insulin pump therapy has been implicated as a risk factor for DKA in the past, most recent studies show that with proper education and practice using the pump, the frequency of DKA is the same for patients on pump and injection therapy.19 Common causes by frequency Other causes Selected drugs that may contribute to diabetic ketoacidosis Infection, particularly pneumonia, urinary tract infection, and sepsis4 Inadequate insulin treatment or noncompliance4 New-onset diabetes4 Cardiovascular disease, particularly myocardial infarction5 Acanthosis nigricans6 Acromegaly7 Arterial thrombosis, including mesenteric and iliac5 Cerebrovascular accident5 Hemochromatosis8 Hyperthyroidism9 Pancreatitis10 Pregnancy11 Atypical antipsychotic agents12 Corticosteroids13 FK50614 Glucagon15 Interferon16 Sympathomimetic agents including albuterol (Ventolin), dopamine (Intropin), dobutamine (Dobutrex), terbutaline (Bricanyl),17 and ritodrine (Yutopar)18 DIFFERENTIAL DIAGNOSIS Three key features of diabetic acidosis are hyperglycemia, ketosis, and acidosis.

267 of Kaplan and Pesce.Initial Evaluation Initial evaluation of patients with DKA includes diagnosis and treatment of precipitating factors (Table 14–18). Calculate the osmolal gap, as described by Equation 14-3, p.Measure the osmolality by both freezing point and vapor pressure osmometers.To the other labled “spike” slowly add 10 \(\mu\)L of the ethanol stock solution while gently vortex-mixing the sample. To one labeled “control”, add 10 \(\mu\)L of distilled water.Place 1 mL of a serum whose osmolality had previously been measured and calculated into each of 2 small glass test tubes.Prepare an ethanol stock solution (75000 \(\mu\)g/mL, 1630 mmol/L) by adding 10 mL of 95% ethanol to 100 mL of distilled water.For those laboratories who have access to both freezing point depression and vapor pressure osmometers, this experiment can demonstrate the effect of a volatile solute on osmolarity.Add a very small volume of this hemolysate for example 10 L, to 1 mL of a sample whose osmolality had previously been measured. Prepare a hemolysate of red blood cells, as described in Exercise #15, Bilirubin.